When your Durable Medical Equipment (DME) business faces government scrutiny, it can threaten reimbursement, enrollment, and even your freedom. CMS, MACs, RACs, UPICs, OIG, and the DOJ actively pursue suspected patterns of DME equipment fraud to protect Medicare and Medicaid funds. What may begin as an administrative audit can quickly escalate into civil or criminal exposure if not handled strategically.
DME Supplier Audits – Why You’re a Target of DME Fraud Enforcement Priorities
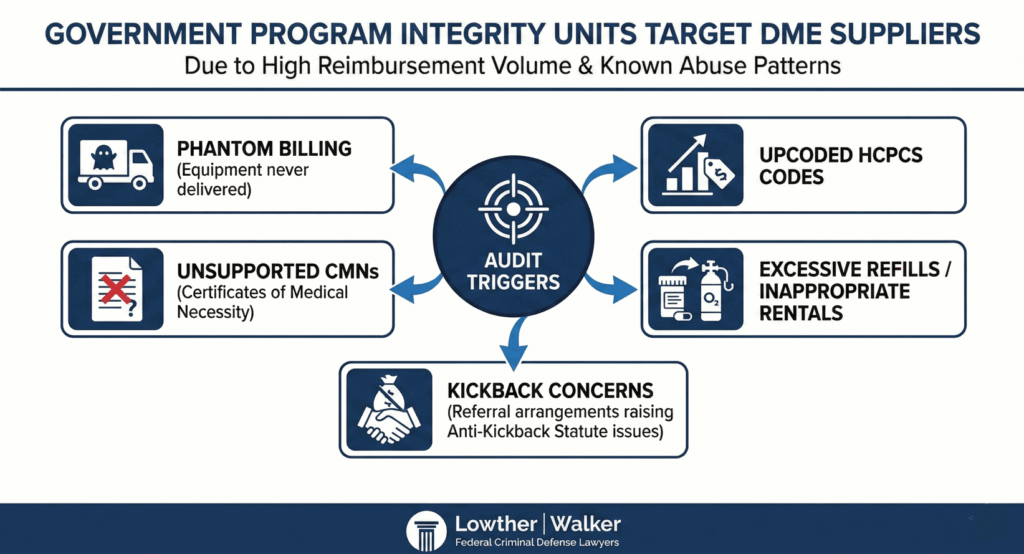
Government program integrity units target DME suppliers due to their high reimbursement volume and known patterns of abuse. Using data analytics and utilization benchmarks, auditors look for:
- Phantom billing (equipment never delivered)
- Upcoded HCPCS codes
- Unsupported Certificates of Medical Necessity (CMNs)
- Excessive refills or inappropriate rentals
- Referral arrangements that raise Anti-Kickback Statute concerns
Even honest documentation errors can be treated as fraud if government data suggests intent. Lowther & Walker
MAC Auditors Make DMEPOS Suppliers a High Priority Target
DMEPOS suppliers are a high-priority target for Medicare audits because the program has consistently high improper payment rates and remains a frequent source of fraud under Medicare Part B.

CMS relies on Medicare-enrolled DMEPOS suppliers to furnish medically necessary equipment and submit claims, often subject to strict requirements, including prior authorization, face-to-face encounters, written orders before delivery, and detailed medical documentation.
CMS reported approximately $1.9 billion in improper DMEPOS payments in FY 2024 alone, representing 21.4% of total DMEPOS spending, prompting aggressive audit and enforcement activity. High-risk product categories such as continuous glucose monitors, orthotic braces, positive airway pressure devices, and surgical dressings have drawn particular scrutiny due to patterns of billing for items not provided, lack of medical necessity, missing or insufficient documentation, unlicensed suppliers, kickbacks, and proof-of-delivery failures.
With improper payment rates exceeding 50% in some categories and well-documented enforcement actions under the False Claims Act, CMS and its contractors continue to treat DMEPOS as a primary audit focus, using audits, prior authorization programs, and post-payment reviews to curb abuse and recover overpayments.
How DME Supplier Government Audits Escalate
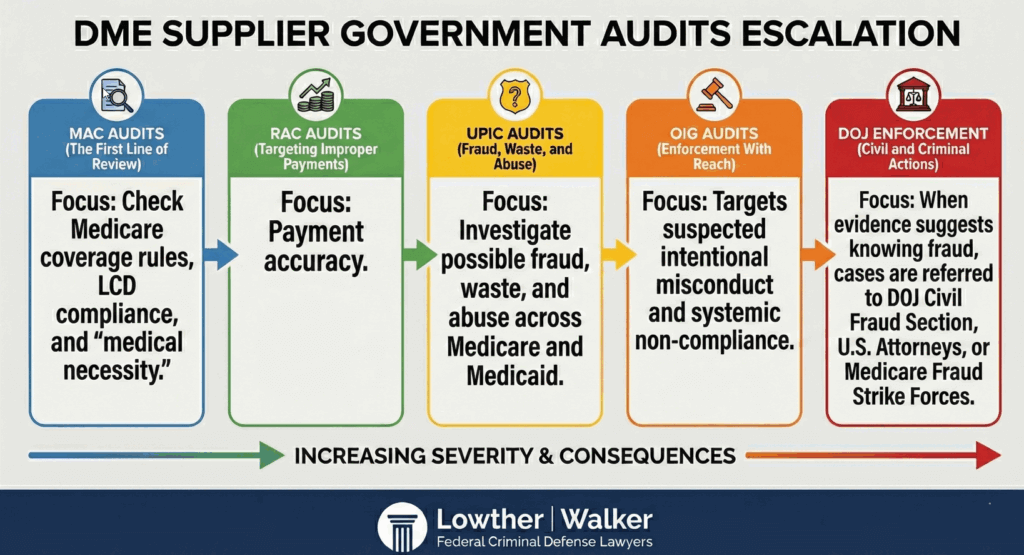
MAC Audits — The First Line of Review
When MAC auditors request documentation, they’re checking Medicare coverage rules, LCD compliance, and “medical necessity.” If discrepancies appear:
Overpayment demands are issued
Recoupment may be calculated across claim pools
Your supplier number could be flagged for pre-payment review
MAC audit findings often feed deeper investigations if patterns persist.
RAC Audits — Targeting Improper Payments
Recovery Audit Contractors focus on payment accuracy:
- Frequency limits
- Same-or-similar service issues
- Policy deviations
Adverse RAC audit findings often lead to larger extrapolated repayment obligations and draw attention from UPICs.
UPIC Audits — Fraud, Waste, and Abuse
Unified Program Integrity Contractors investigate possible fraud, waste, and abuse across Medicare and Medicaid. UPIC involvement can mean:
Payment suspension
Denial of future claims
Referral to DOJ or State Medicaid Fraud Control Units (MFCUs)
At this stage, UPIC audit responses must be handled with precise legal strategy.
OIG Audits — Enforcement With Reach
The HHS Office of Inspector General targets suspected intentional misconduct and systemic non-compliance. Possible outcomes include:
- Civil Monetary Penalties (CMPs)
- Program exclusion under 42 U.S.C. §1320a-7
- Corporate Integrity Agreements (CIAs)
OIG findings often underpin False Claims Act cases against suppliers.
DOJ Enforcement — Civil and Criminal Actions
When evidence suggests fraud, cases are referred to the DOJ Civil Fraud Section, U.S. Attorneys’ Offices, or Medicare Fraud Strike Forces. Penalties can include:
- False Claims Act liability with treble damages
- Statutory fines per false claim
- Criminal healthcare fraud charges
- Conspiracy or kickback prosecutions
Some cases involve parallel civil and criminal tracks. Early defense involvement shapes outcomes.
DME Fraud Audit Defense Case Example
United States v. S.I. — Telemedicine Kickback / DME Fraud
The HHS-OIG investigated a provider for a $30 million telemedicine kickback scheme involving unnecessary DME and genetic testing reimbursements.
Lowther | Walker’s defense team conducted an internal investigation, engaged with auditors and prosecutors, and convinced the government not to prosecute the client.
This outcome preserved the client’s liberty and their practice.
Case summary: Strategic early engagement and evidence management can prevent prosecution even in large alleged fraud schemes.
When a “Routine Healthcare Audit” Is Not Routine
If you’ve received notice that the government will be conducting a routine audit of your business records, now is the time to consider pre-emptive legal defensive action. Once audits begin and record requests occur, the potential for penalties increases.
The following four scenarios indicate a high-risk audit:
- Requests expand beyond simple documentation
- UPIC or OIG involvement is indicated
- Payment suspension threats arise
- Government agents ask probing intent questions
Request a Free DME Healthcare Audit Consultation with Lowther | Walker
With more than 20 years of experience responding to government audit requests, Lowther | Walker brings a proven, strategic approach to healthcare audit defense. Our attorneys understand how to control audit communications, challenge improper extrapolation methodologies, and build strong medical necessity defenses while proactively mitigating criminal exposure.
We regularly negotiate and communicate directly with auditors and federal prosecutors. Our attorneys can preserve DME supplier program enrollment and billing rights and coordinate defense strategies across parallel civil, criminal, and administrative proceedings.
If your business is facing an audit or federal investigation, contact Lowther | Walker immediately by phone at 877-208-7146 or by booking your free consultation to protect your operations, your reputation, and your future.
FAQs for DME Suppliers Facing Healthcare Fraud Allegations
What actions often trigger a fraud investigation?
Abnormal billing volumes, inconsistent documentation, referrals tied to financial interests, or adverse audit patterns.
Can an administrative audit lead to criminal charges?
Yes — UPIC and OIG audits frequently precede referrals to DOJ if intent or fraud patterns emerge.
Are honest mistakes treated as fraud?
If patterns suggest systemic issues or a lack of controls, billing mistakes can be framed as knowing conduct under federal laws.
What penalties can suppliers face?
Recoupment, CMPs, exclusion from federal programs, treble damages under the False Claims Act, fines, and imprisonment.
Why is early legal representation important during DME audits?
Early counsel can limit extrapolation, reduce audit escalation, and influence government charging decisions.
Do audit case outcomes vary widely?
Yes — from non-prosecution and probation to complete acquittals, strategic defense significantly impacts results.